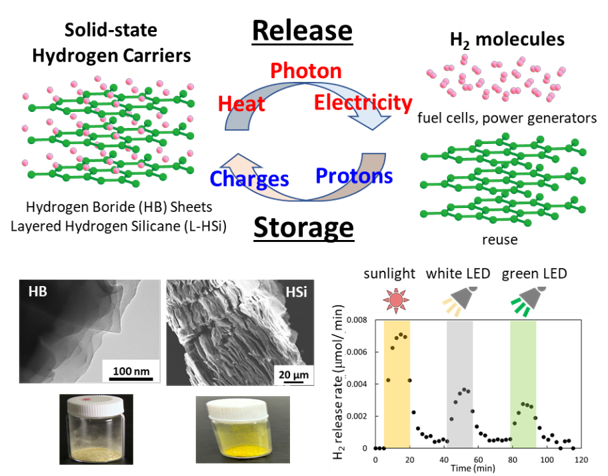
Toward a carbon-neutral society, the widespread use of hydrogen energy requires the development of hydrogen carriers that can safely store and transport hydrogen and release it with low energy input. High-pressure hydrogen gas cylinders, which are commonly used as hydrogen carriers, have low hydrogen storage density and pose risks of explosion. Liquid hydrogen carriers such as ammonia, formic acid, and organic hydrides also present challenges, including toxicity and corrosiveness, as well as the need for high-temperature heating to release hydrogen. In addition, solid hydrogen carriers such as metal alloys have low gravimetric hydrogen density, making lightweight storage and transportation difficult. In our laboratory, we are studying nanosheet-based two-dimensional materials that contain large amounts of hydrogen.
As hydrogen-containing two-dimensional materials, we are studying hydrogen boride sheets (HB sheets), and layered hydrogenated silicene (L-HSi). These materials exhibit extremely high gravimetric hydrogen density (8.5% in the case of HB sheets), are lightweight, and do not pose a risk of explosion. In addition, they can be synthesized by a facile solution-based method, making large-scale production feasible. In our laboratory, we aim to achieve energy-efficient hydrogen release by applying various external stimuli (such as heat, photon, and electricity) to these materials, and we are also working on re-storage of hydrogen after release. Our goal is to realize lightweight, safe, and energy-efficient hydrogen carriers.